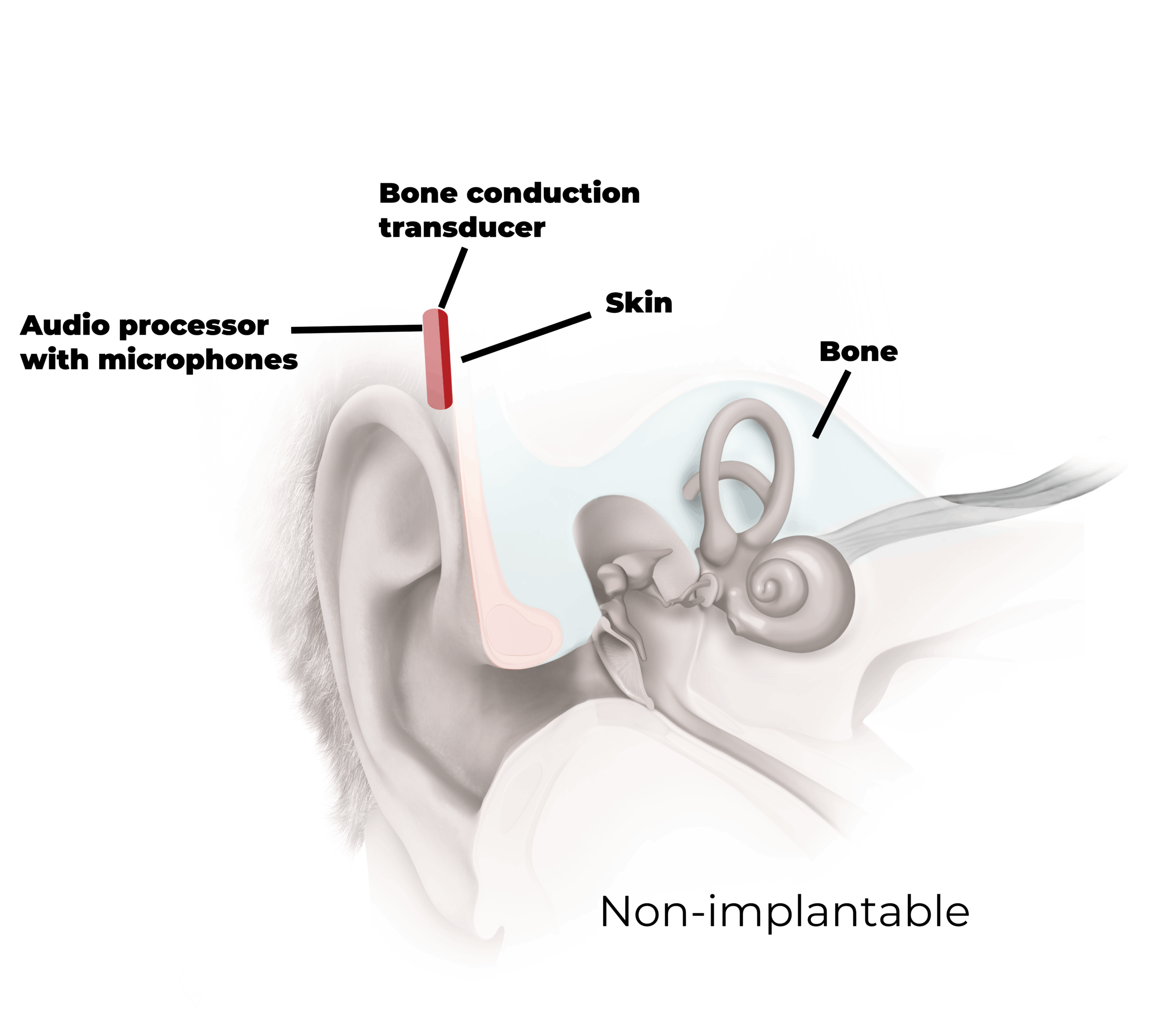
Non-implantable
This section outlines the benefits and drawbacks of a non-implantable solution, categorized into three key domains: audiological aspect, medical aspect, and patient-reported aspects.
Each publication listed below is rated by its importance for patients, using a Hearring key icon scale 🔑— one key for low importance, three keys for medium importance, and five keys for high importance.
🔑 low importance for patients
🔑🔑🔑🔑🔑the highest importance for patients
Benefits
Audiological Aspects
🔑🔑🔑🔑
1. “Users of the passive [bone conduction] implant received comparable hearing benefit with the ADHEAR. The mean aided thresholds in sound field measurements and speech understanding in quiet and noise were similar, when subjects were evaluated either with the ADHEAR or the passive [bone conduction] implant. The audiological outcomes for the non-implanted group were also comparable between the ADHEAR and the [bone conduction device] on softband.” (Skarzynski et al. 2019)
2. In two studies by Gawliczek et al. the ADHEAR showed the same audiological performance as BAHA5 on SoundArc, but has the advantage of superior wearing comfort due to the lack of pressure. (conclusion / Gawliczek et al. 2018)
3. “The mean unaided free field hearing threshold of 508B HL(.), improved significantly by 228B (13.0-29.9) with the
ADHEAR and by 236B (13.6-32.9) with the [bone conduction hearing aids] (p&O.001). Furthermore, the mean unaided speech recognition threshold (SRT) in quiet improved significantly by 198B (10.3-28.1) with the ADHEAR and by 21AB (12.6-29.4) with the [bone conduction hearing aids] (p@0.001). For both audiological tests, there were no significant differences between the ADHEAR and the [bone conduction hearing aids].” (Favoreel et al. 2020)
4. “The mean aided threshold of the 12 children with Baha Softband is 27 dBHL ₫6 dBHL. In the reference group, with the conventional bone conductor, this is 25 dBHL ÷6 dBHL.” (Verhagen et al. 2008)
More: Gavilán et al. 2019, Zernotti et al. 2019, Brill et al. 2019, Dahm et al. 2019, Weiss et al. 2019, Neumann et al. 2019, Urik et al. 2019, Dahm et al. 2018, Hol et al. 2005
Medical Aspects
🔑🔑🔑🔑🔑
1. As non implantable devices are non-invasive and easy to use, every group of age can benefit from this treatment option.
(HEARRING consensus)
2. “For very young children with congenital bilateral aural atresia, in whom surgical intervention is not an option (for the time being), bone conduction hearing aids are the recommended method of hearing rehabilitation.” (Verhagen et al. 2008)
🔑🔑🔑🔑🔑
1. As non-implantable devices are always attached to the skin via an external fixation, the device can be taken off whenever necessary or wanted. (HEARRING consensus)
2. “The use of [bone anchored hearing aids) on a softband (elastic fabric) or headband (diadem type) (…) is the gold standard for preoperative assessment. ” (Reinfeldt et al. 2015)
3. “The headband or test rod, although crude in many ways, still offers an easy, fast and objective way of assessing bone conduction capacity which allows the patient to directly experience bone conducted sound, and so helps in decision making for him/her.” (Hagr et al. 2007)
4. It is possible to predict the hearing outcomes of an active [bone conduction] implant through the pre op use of a non implantable device (Adhear) (conclusion / Canale et al. 2019)
5. “This adhesive hearing system is a safe and effective device to treat transient conductive hearing loss and may considerably improve treatment for patients even with short-term hearing loss.” (Weiss et al. 2019)
More: Spielmann et al.2018, Neumann et al. 2019, Mertens et al. 2018, Osborne et al. 2019, Zarowski et al. 2011
🔑🔑🔑🔑
1. As non-implantable devices are always attached to the skin via an external fixation, the device can be taken off whenever necessary or wanted. (consensus)
More: Neumann et al. 2019, Mertens et al. 2016, Osborne et al. 2019
Patient-reported Aspects
🔑🔑🔑🔑
1. The simple removal of the external device is the only necessity to undergo a MRI. (HEARRING consensus)
Drawbacks
Audiological Aspects
🔑🔑🔑🔑
1. “Adapter adhesion was sufficient for most of the subjects in our study but varied between one and seven days among participants. Excessive handling of the adapter or rubbing against car seats, armrests, hats etc. as well as variations in mastoid shape, skin type, sweating, or a combination of these factors could cause the observed differences.” (Neumann et al. 2019)
🔑🔑🔑🔑
1. “Observation in clinical practice shows that, still, the majority of [bone conduction device] trial experiences in the general SSD population are negative.” (Mertens et al. 2018)
Medical Aspects
🔑🔑🔑🔑
1. “In conclusion, this new, adhesive bone conduction hearing aid has a high patient satisfaction rate while causing no skin irritation or pain.” (Dahm et al. 2019)
2. “Bone conduction hearing devices integrated in softbands (BCDSs) are frequently not well accepted by children with conductive hearing loss due to pressure on the head, sweating, or cosmetic stigma.” (Neumann et al. 2019)
More: Verhagen et al. 2008, Osborne et al. 2019, Canale et al. 2019, Reinfeldt et al. 2015
Patient-reported Aspects
🔑🔑🔑🔑
1. “Electrical Stimulation via a Cl resulted in a significant reduction in tinnitus loudness (…).” (Van de Heyning et al. 2008)
More: Häußler et al. 2019, Liu et al. 2018, Dillon et al. 2017, Tavora-Vieira 2015, Mertens et al. 2015, 2016, Peter et al. 2019

